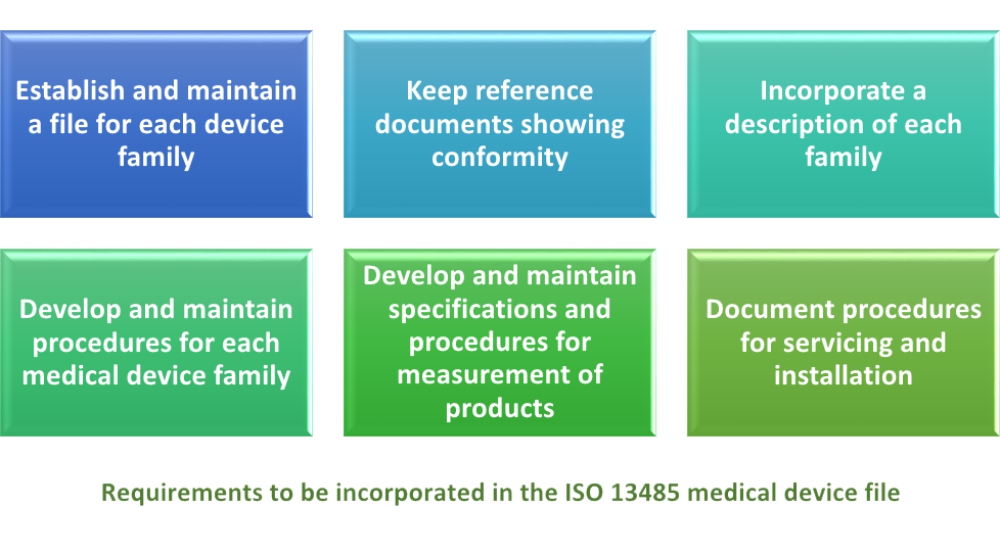
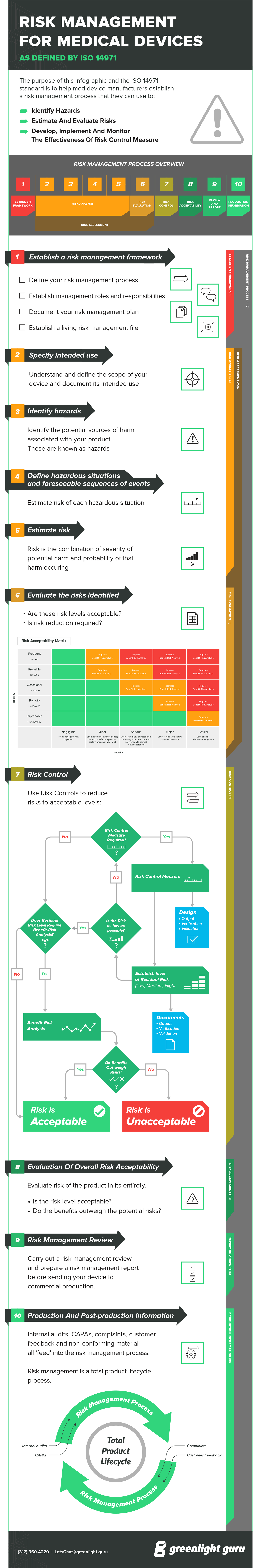
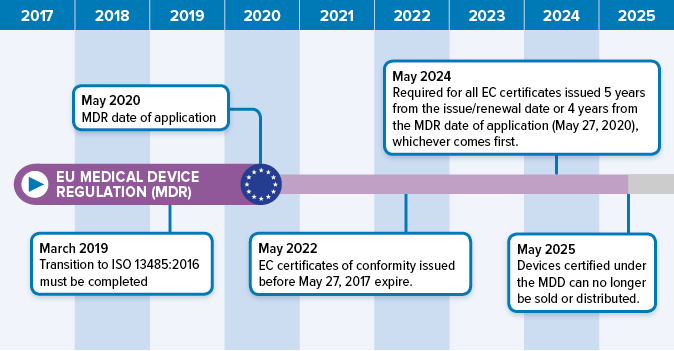
Requirements of iso 13485 2016 are applicable to organizations regardless of their size and regardless of their type except where explicitly stated.
Iso medical device labeling standards.
Sign up to our newsletter for the latest news views and product information.
An interlaboratory comparison of analytical methods for ethylene oxide pb 86.
Keep up to date with iso.
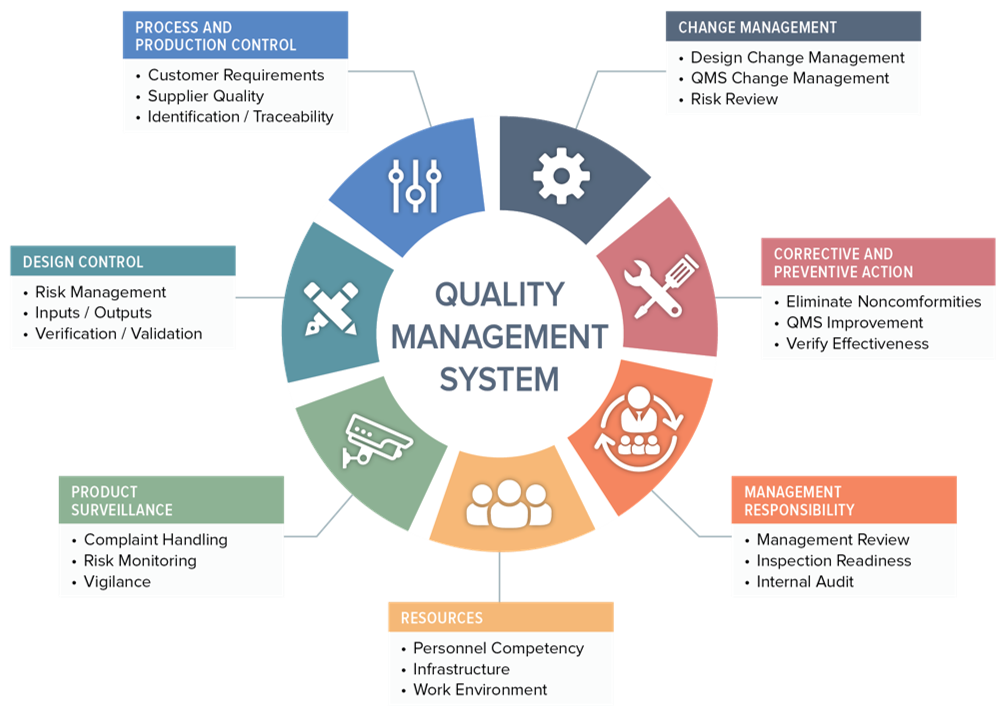
Iso 13485 medical devices quality management systems requirements for regulatory purposes is an international organization for standardization iso standard published for the first time in 1996.
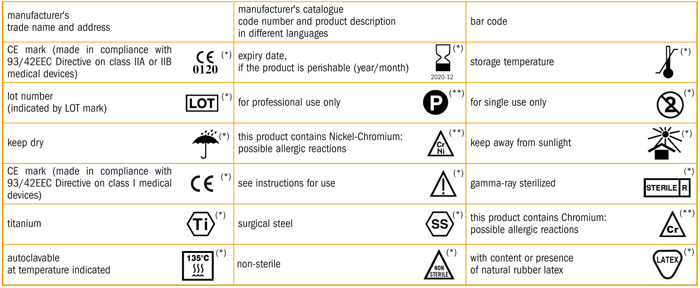
It also lists symbols that satisfy the requirements of this document.
The iso 13485 standard was entitled quality systems medical devices.
This article explains the method starting with standards from the international organization for standardization iso adopted and recognized in various regulatory systems.
The iso 9001 standard was entitled quality systems model for quality assurance in design development production installation and servicing.
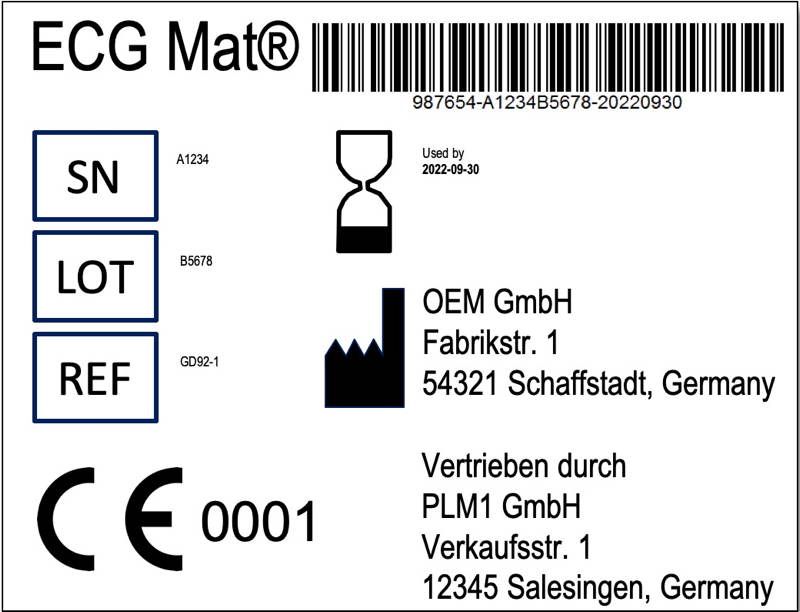
Iso 15223 1 2016 identifies requirements for symbols used in medical device labelling that convey information on the safe and effective use of medical devices.
Medical devices information to be supplied by the manufacturer.
The primary standards included international organization for standards iso 9001 1994 and 13485 1996.
It represents the requirements for a comprehensive quality management system for the design and manufacture of medical devices this standard supersedes earlier documents such as en 46001 1993.
The article uses iso 13485 2003 and iso 14971 2007 as illustrations p.
Iso 13485 is the quality management system standard accepted as the basis for ce marking medical devices under european directives and regulations.
Iso 13485 is the best internationally accepted model a medical device organization can implement to help demonstrate compliance to laws and regulations of the medical device industry.
Wherever requirements are specified as applying to medical devices the requirements apply equally to associated services as supplied by the organization.
P one common source of misunderstanding in the medical device industry is the method the various national regulatory systems use to identify standards.